All about plutonium
What is plutonium?
Plutonium is a chemical element with the symbol Pu and the atomic number 94. It is a hard, white metal that resembles iron. The forms of plutonium most commonly used in the nuclear industry are Pu 239, Pu 240 and Pu 241.
Where does plutonium 239 come from?
Originally, Pu 239 was formed during the creation of the universe!
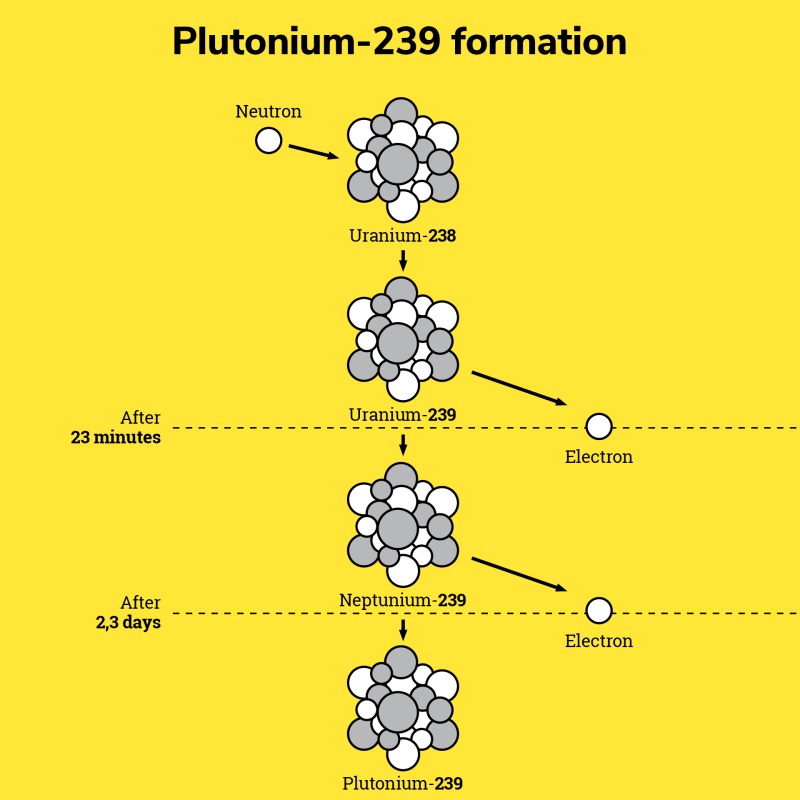
So how the nuclear industry can use Pu 239, you may wonder… The answer is simple: it is produced by the nuclear reaction from the uranium used as fuel. In this reaction, uranium-238
captures a neutron and transforms into uranium-239. In turn, the uranium-239 converts to neptunium-239 by losing an electron. Then, in the same way, the neptunium-239 transforms into plutonium-239. After a mechanical and chemical treatment of the
used fuel in a specialized plant (Orano la Hague plant), the recovered plutonium is recycled in nitrate form and then converted into the oxide (PuO2).
- Uranium 238 : 92 protons 146 neutrons
- Uranium 239 : 92 protons 147 neutrons
- Neptunium 239 : 93 protons 146 neutrons
- Plutonium 239 : 94 protons 145 neutrons
What is plutonium used for?
Plutonium has several industrial uses, particularly in the nuclear industry.
Plutonium-239 is primarily used as a fuel to power nuclear reactors. In fact, it enters into the composition of MOX fuel – mixed uranium and plutonium oxide. By combining the Pu 239 produced by nuclear reactors with depleted uranium, MOX can be used to fabricate one new fuel assembly from every 8 used fuel assemblies. The use of this fuel also presents the great benefit of dividing by 5 the amount of final waste generated by the nuclear industry and offering a solution for dealing with some of the issues around plutonium. MOX is exclusively manufactured by Orano on the Melox site. Since 1972, MOX fuel assemblies have been used by 44 nuclear reactors around the world.
Plutonuim-238, on the other hand, has significant spatio-temporal properties. It is used to power space probes sent far into space, beyond our solar system. These space probes are also equipped with solar panels but the further they move away from the sun, the less light there is and the less effective the panels are. This is where Pu 238 comes in. As plutonium-238 naturally self-heats through its radioactivity, it can be used to operate what are referred to as Radioisotope Thermoelectric Generators (RTG). These are a kind of super battery able to withstand the extreme conditions imposed by space. The Pioneer 10, Pioneer 11, Voyager 1 and 2, and Galileo probes are equipped with such devices.
A little anecdote
By way of analogy with the earlier discoveries, uranium and neptunium – whose names owe their origin to the planets Uranus and Neptune – the name plutonium, given to the element in 1942, is a reference to the planet Pluto, situated beyond Uranus in our solar system.
- Understanding nuclear energy Nuclear energy makes it possible to produce low-carbon, competitive, and continuous electricity
- Safety at work: a major priority for the nuclear industry Laurence Gazagnes and Mounir Boukil discuss the risks inherent in plant operation and maintenance. With results to back up their points of view!
- All about radioactivity Radioactivity is part of the universe. It is present everywhere, both in the atmosphere and in the earth's crust, which contains radioactive elements.